Histology Lab Test2/11/2021
Employees must become educated to package and ship according to the rules and in a manner that matches to their function-specific obligations.Some content material may become in British until all the content material can be translated.Timely conversation between medical and laboratory employees is essential to reduce the risk incurred in handling individuals from patients with feasible SARS-CoV-2 an infection.Such individuals should be labeled accordingly, and the laboratory should become notified to make certain correct specimen managing.
Common and specific biosafety suggestions for handling SARS-CoV-2 specimens are offered below. For extra information on handling SARS-CoV-2 specimens, send to the Lab Frequently Requested Questions. Anatomic pathology contains surgical pathology, histotechnology, cytoIogy, and autopsy. Risks are improved in the operative grossing room during regular specimen handling, tissues dissection, and the planning of frosty sections of tissues making use of a cryostat. 
See Laboratory Frequently Questioned Questions for even more information. Clinical pathology uses different procedures and workflows than those utilized in anatomic pathology, and consequently the dangers and mitigation settings required to shield personnel are different. At a minimum amount, all personnelwhether practicing anatomic or clinical pathologyshould follow Regular Safety measures when dealing with patient tissue and specimens. 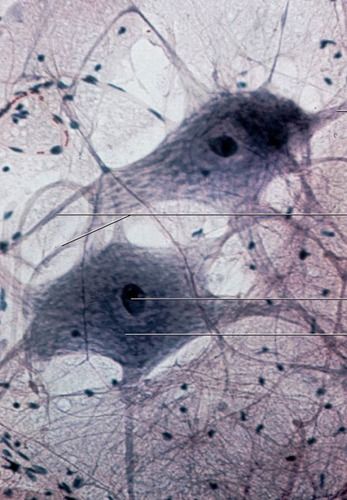
Testing websites that function a POC diagnostic device must possess a present Clinical Lab Improvement Changes of 1988 (CLIA) certificate. During the COVID-19 open public health crisis, the Facilities for Medicare Medicaid Providers (CMS) will permit a laboratory to prolong its present Certificate of Waiver to function a short-term COVID-19 assessment web site in an off-site area, like as a long-term care facility. The temporary COVID-19 screening site is only allowed to carry out waived checks, consistent with the laboratorys existing certification, and must end up being under the direction of the present lab director. If needed, additional safety measures can become used, like as a operative face mask or face guard, or various other physical obstacles, like as a splash shield to function behind. Pursuing the producers recommendations for use, such as dilution, contact period, and safe handling. Illustrations of these additional precautions include personal protective apparatus (PPE), like as a operative cover up or face cover, or some other physical obstacles, like a dash cover; centrifuge protection cups; and covered centrifuge rotors to reduce the risk of exposure to laboratory personnel. The donning and doffing area should not really end up being in the work area. Web site- and activity-specific biosafety danger examination should become carried out to figure out if extra biosafety precautions are warranted structured on situational requirements, like as higher testing quantities or large quantities, and the possibility to create infectious minute droplets and aerosols. To determine appropriate biosafety mitigation procedures, laboratories should carry out an activity-specific biosafety danger assessment that evaluates lab facilities, employees and training, procedures and techniques, safety equipment, and risk mitigation methods. Biosafety specialists, laboratory management, and medical and protection experts should be involved in the danger assessment procedure. Presently, there is usually no proof to suggest that this laboratory waste wants any extra product packaging or disinfection processes.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
